
TruSight Oncology 500 (TSO500)
Pan-cancer next-generation sequencing assay for biomarker identification and comprehensive genomic profiling across 523 genes
TruSight Oncology 500 (TSO500)
Pan-cancer next-generation sequencing assay for biomarker identification and comprehensive genomic profiling across 523 genes

Clinical Genomics Laboratory Services
Next Generation Sequencing Services
Microsatellite Instability Assay
Immune Landscape Signatures
Oncomine Precision Assay
Tumor Mutational Burden
TruSight Oncology 500 (TSO500)
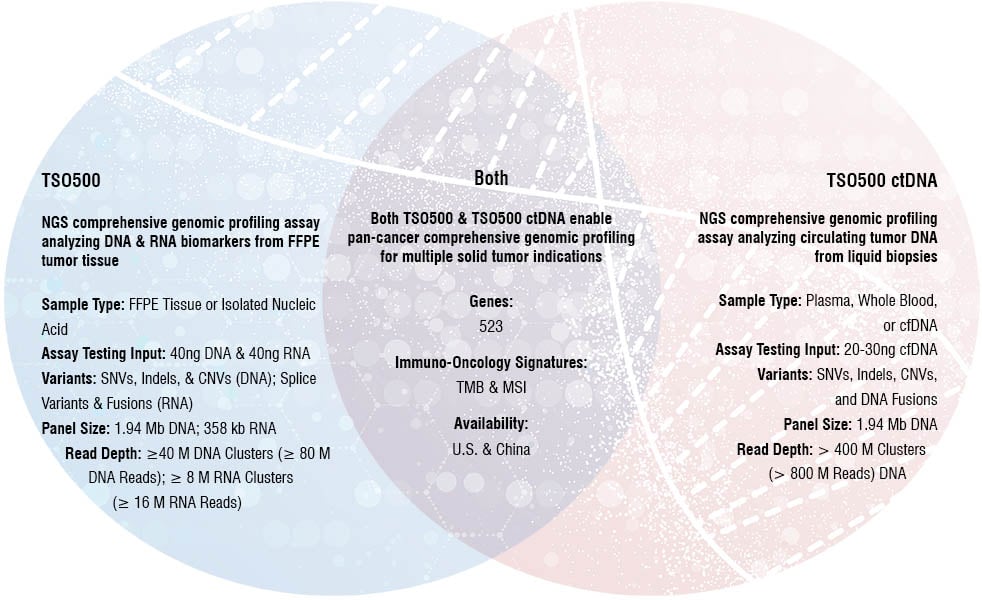
TruSight Oncology 500 ctDNA
Whole Exome Sequencing
Liquid Biopsy ctDNA
TruSight Oncology 500 ctDNA
Single Cell and Spatial Genomics
Single Cell RNA Sequencing
Spatial Genomics
Biomarker Discovery
Technology Platforms
Bioinformatics Services
Ordering and Client Login
Central Laboratory Services
A Hybrid-Capture Approach for Target Enrichment of 523 Genes Across Various Tumor Types
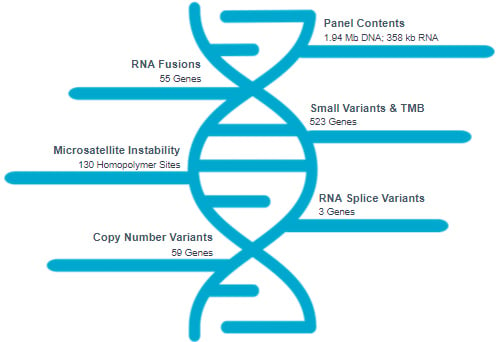
Gain an expansive view of your solid tumor samples by targeting 523 cancer-related genes using the TruSight Oncology 500 assay. Not only does this assay identify all relevant variants (SNVs, insertions, deletions, CNVs, gene fusions, and transcript variants) across various tumor types, but it also reports tumor mutational burden (TMB) and microsatellite instability (MSI) scores.
Our comprehensive genomic panel has advantages over individual biomarker assays in its ability to detect multiple biomarkers with one test, saving time, money, and samples. Q2 Solutions can meet your profiling needs by leveraging trusted technology from Illumina® in combination with our global laboratory network and expert support team.
An Efficient, Reliable Solution for Tumor Characterization
Minimal input, extensive results
Obtain full exon coverage of 522 out of 523 genes using as little as 40 ng of DNA and 40 ng of RNA input. TSO500 can detect biomarkers without the need of a matched normal sample.
Simple, cost-effective workflow
Detect multiple biomarkers with a single assay to save time, money, and samples. The distributable kit enables global implementation.
Confident variant detection
Obtain a high-resolution view of variants enabled by TSO500’s hybrid-capture chemistry and unique molecular indices (UMIs).
Measurement of key immuno-oncology biomarkers
Gain insight into TMB score and MSI score in addition to variant calls. TruSight Oncology 500 uses an enhanced algorithm to robustly assess TMB from SNVs and indels detected in coding regions.
Expansive services enabled by our global network
Our centralized, scientific operational oversight combined with our global laboratory footprint enable comprehensive, large-scale trial support. We enable clinical development support irrespective of geography.
Flexible customization to satisfy specific needs
We work with you to understand your specific needs, and our team provides cost-effective recommendations from custom assay development to specialized bioinformatics support.
TSO500 Variant Detection
TSO500 Assay Comparison
Deep Sequencing Enabled by Q2 Solutions’ Subject Matter Expertise
Feel confident in your results with the trusted Illumina® software pipeline. Our software teams validate the most up-to-date software to ensure you are receiving optimal results. Q2 Solutions can develop custom reporting and filtering to meet your specific research needs. Our technical support teams help you understand your deep sequencing data with our superior bioinformatics knowledge.

Deliverables
Q2 Solutions provides a text-delimited, combined variant output file for all biomarkers
- QC Metrics
- CNV Outputs
- RNA Splice Variants
- RNA Fusion Report
- Genomic VCFs (SNVs & Indels)
- Variant Call Annotations
- MSI Scores
- TMB Scores
- FASTQs
- BAMs
Regulatory
Validated up to CAP/CLIA standards
Q2 Solutions offers this TSO500 assay at multiple regulatory levels, from Research Use Only (RUO) to Good Clinical Practices (GCP) to CAP/CLIA, depending on the intended use of the assay by our client. Please reach out to our Q2 Solutions Genomics Business Development team to discuss your intended use so we can better guide which regulatory level is most appropriate for your clinical or research program(s).
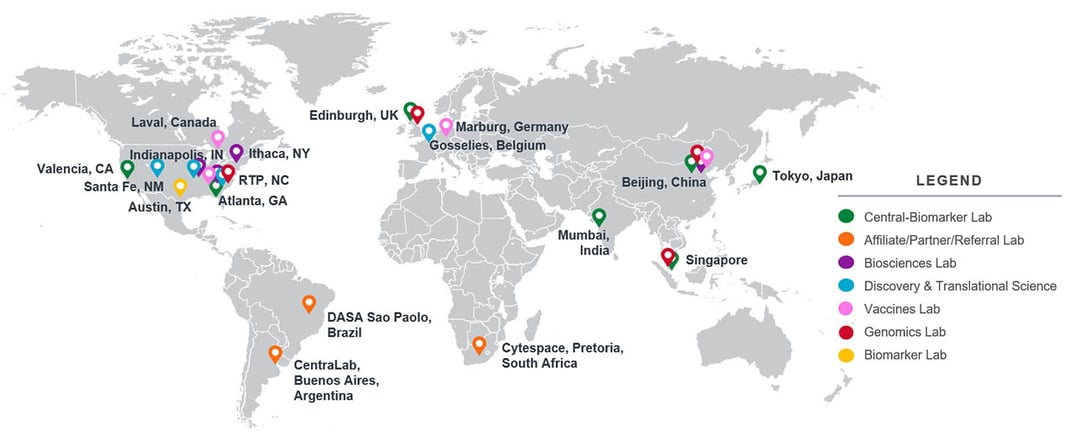
Supporting Studies Around the World
Our centralized, scientific, and operational oversight combined with our global laboratory footprint enables comprehensive, large-scale trial support. You will be supported every step of the way by our scientific project management team who will work with you to understand the most cost-effective recommendations to meet your specific needs. We are committed to transparency so you can feel confident in our promises of quality and timely delivery. We can expert support team can meet your research needs from nearly anywhere in the world with our genomic centers in the United States, China, United Kingdom, and Singapore.
Learn More About TSO500 with Q2 Solutions
Posters
- TMB Standardization by Alignment to Reference Standards
- Panel-based TMB Analysis of Matched Tumor and Plasma Specimens Using Illumina’s TSO500 NGS Assay
- Comprehensive Genomic Profiling in FFPE Tissue Sample using TruSight Oncology 500 Assay
Presentations
- Approaches to Demonstrating Commutability of Various NGS Reference Standards for Targeted ctDNA Analysis
- Analytical Validation of Illumina’s TSO500 ctDNA Assay
Whitepaper
Fact Sheets
Webinars
- Bridging the Gap in Liquid Biopsies with CTCs and ctDNA
- Comprehensive Genomic Profiling from Liquid Biopsy Samples: Clinical Trial Considerations
Podcasts
- The Standardization of TMB by Alignment to Reference Standards
- Liquid Biopsy Collection in Clinical Development Programs
Press Release
Video
Simplify Your Work

Request a Quote
Request a quote today to discover how TSO500 with
Q2 Solutions can advance your research.
Q2 Solutions can advance your research.
Ship Samples
Ship your samples to one of our global genomic laboratory centers.
Get Results
Obtain in-depth data revealing insights into your solid tumor samples.
Related Services & Solutions
Related Thought Leaders Insights
TruSight Oncology 500 (TSO500) Tissue Assay
The TruSight Oncology 500 (TSO500) for tissue assay employs a hybrid capture-based approach targeting 523 genes and leverages unique molecular indices to enable detection of low allele frequency...
Panel-based tumor mutational burden (TMB) analysis of matched tumor and plasma specimens using Illumina’s TruSight Oncology 500 next-generation sequencing assay
Checkpoint inhibitor (CPI) therapy demonstrates a remarkable clinical benefit in many cancer types. However, the ability to successfully select patients who will benefit from CPIs is still limited....
TMB Standardization by Alignment to Reference Standards
At ASCO 2019, we presented a poster with Friends of Cancer Research (FOCR), highlighting their Tumor Mutational Burden (TMB) Harmonization Project. The goal of this Project is to harmonize TMB...














