In Vitro Immunology
In Vitro Immunology

Discovery Sciences
Protein Sciences
Antibody Discovery
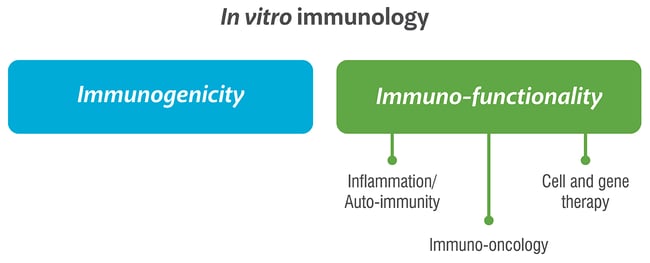
In Vitro Immunology
Immunogenicity
Immuno-functionality
Our Approach
Consultative services
Additional Specialty Testing Services
Watch a webinar to learn how our ImmunXperts team can help you assess the immunogenicity risks of your biotherapeutics.
ImmunXperts, a Q2 Solutions Company, provides trusted immunogenicity risk assessment services, including in silico and in vitro methods, using T-cell activation and proliferation as a surrogate marker for the induction of anti-drug antibodies, cytokine release assays, and MHC-associated peptide proteomics (MAPPS). Our methods provide functional insights into immune-mediated adverse events.
ImmunXperts also performs functional screening assays for drug candidates in immuno-oncology, cell and gene therapy and other disease areas. Our consultative services provide theoretical immunogenicity and immunology courses, protocol review and optimization, and hands-on training, including training on optimal procedures for the isolation and cryopreservation of PBMCs from whole blood.
Located in Gosselies, Belgium, our ImmunXperts team assists clients in advancing programs from discovery to pre-IND and exploratory clinical stages. We bring broad experience helping clients address unique challenges across all molecule types. Our team is experienced with an array of new modalities such as novel anti-oncolytic molecules, cell and gene therapy products, monoclonal antibodies (mAb) bispecific antibodies, new antibody formats, small molecules, and generic peptides.
ImmunXperts also hold a Belgian Biobank License and store high-quality PBMC samples from more than 1300 healthy donors with 4-digit HLA typing to ensure appropriate representation of diverse global populations.