Immunogenicity
Immunogenicity

Discovery Sciences
Protein Sciences
Antibody Discovery
In Vitro Immunology
Immunogenicity
Immuno-functionality
Our Approach
Consultative services
Additional Specialty Testing Services
Watch a webinar to learn how our ImmunXperts team can help you assess the immunogenicity risks of your biotherapeutics.
ImmunXperts’ immunogenicity testing allows you to de-risk your pipeline and optimize candidates for reduced adverse events and lower immunogenicity risk. Choose from a comprehensive immunogenicity assessment or just the components you need.
In silico methods for low-cost, high-throughput assessment
- Computer-based assessment of T-cell epitopes for HLA-binding risk
- Sequence-based assays enable rapid assessment of large numbers of lead candidates
- Useful for deimmunization (lowering unwanted immunogenicity)
In vitro early/innate assays
Our in vitro assays include flow cytometry, cytokine profiles, and ELISpot options
- Cytokine release assay – using whole blood or PBMCs (human and NHP); customizable up to 60 cytokines
- Monocyte activation assay
- Dendritic cell (DC) activation/maturation assay
- Reporter cell line assay
In vitro T-cell assays
- CD8-depleted PMBC assay – T-cell proliferation by flow cytometry
- DC/T-cell assay – appropriate for immunomodulatory molecules known to directly interact with T cells
- Peptide assay – ultrasensitive Fluorospot assay; customizable up to 4 cytokines
MHC-associated peptide proteomics (MAPPS Assay)
- Highly specialized technique using mass spectrometry to determine which peptides are presented on the cell surface on MHC molecules; often followed by a peptide assay to evaluate T-cell response and identify the immunodominant epitopes
Anti-drug antibody (ADA) assay
- Evaluates pre-existing anti-drug antibodies present in global or specific populations
Immunogenicity: What is immunogenicity of therapeutics? Why is it important?
Protein therapeutics and cell and gene therapies represent a significant proportion of the currently available therapies for a broad spectrum of diseases and is one of the fastest growing markets. However, the development of these biotherapeutics can be challenging.
One major hurdle in the therapeutic development is the risk for unwanted immunogenicity—the ability to trigger an unwanted immune response which translates in the clinic to anti-drug antibodies, which can lead to decreased efficacy and safety concerns.
Today, several tools are available to improve and accelerate therapeutic drug development in an early stage. A well balanced and rational development strategy can help to deselect immunogenic drugs early on and reduce the number of clinical failures.
As the in vivo induction of anti-drug antibodies is a complex process, a multistep approach or a combination of tools might be required to document this risk in a non-clinical set-up.
Often used as a first step is an in silico T-cell epitope prediction algorithm such as NetMHCIIpan, which can be used to assess and compare the immunogenic risk potential of the lead candidates based on the primary sequence and guide de-immunization strategies.
Further monitoring of the innate immune response can be done with cytokine release assays. Assessment of the immunogenic risk can be performed using the MAPPs (MHC-associated peptide proteomics) assay and in vitro T-cell proliferation and activation assays to determine and rank the immunogenic risk of the test proteins or identify specific regions of concern.
To manage this unwanted immunogenicity, in vitro assays using human primary immune cells can be used to assess both the innate as well as the adaptive response. To achieve reliable and consistent results, high quality primary immune cells should be used in combination with sensitive fit-for-purpose in vitro assays.
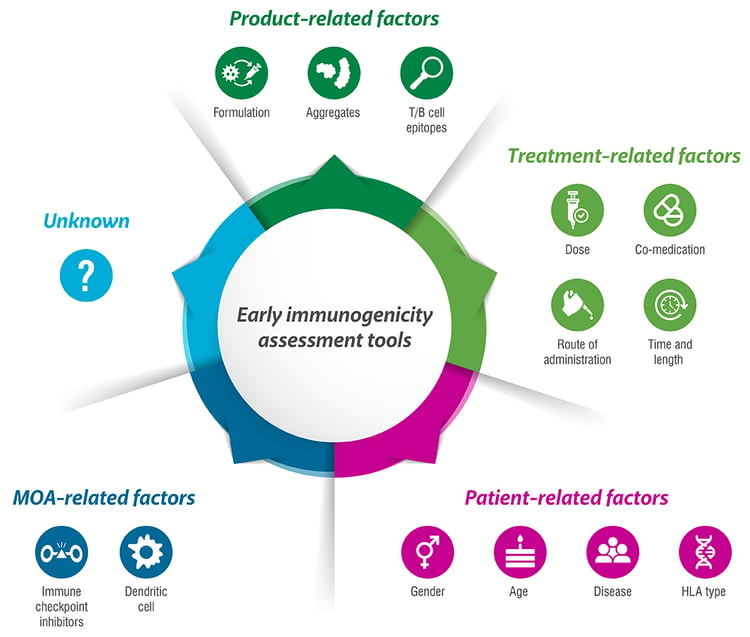
Which factors can affect immunogenicity?
Unwanted immunogenicity can be triggered by many different factors, which can be divided in several categories. These include product-related factors, treatment-related factors, patient-related factors, and mode of action.
Product-related factors comprise:
- The primary sequence, which can be recognized as foreign by the immune system, even if the protein was humanized or even fully human;
- Aggregates, which can be there from the manufacturing process on or which can be induced over time or by handling due to agitation or heat; and
- The formulation, which can trigger an innate immune response and thereby enhance the adaptive response, or which can influence the molecule.
Treatment-related factors are:
- The dose of the therapeutic, where a higher dose does not necessarily mean a higher chance for an unwanted immune response;
- Co-medication, which could decrease (such methotrexate) or increase (observed for certain combinations of oncologic biotherapeutics) the chance of ADA-formation in the clinic;
- The route of administration, although there is not one that is in general more immunogenic compared to the other, it remains therapy- and disease specific; and
- The time and duration of the therapy, where a short administration can induce a different immune response compared to a repeated lengthy therapy.
Patient-related factors can contribute to the immunogenic outcome of the therapy:
- The HLA type is a determining factor as the MHC molecules are decisive for what is presented to the cells of the adaptive immune system;
- The disease for which the therapy is given, as the immune response toward the biotherapeutic might differ between an inflammatory, auto-immune, oncologic, or other diseased environment;
- The age of the patient plays a role, as a young person has a different distribution of naïve versus memory T cells compared to an older person; and
- The gender of a person might lead to a different outcome, due to the influence of the endocrine system on the immune system.
With the newer biotherapeutics, the mode of action (MOA) has also proved to be of great influence, with molecules targeting the immune system showing an (inhibitory or stimulatory) risk of immunogenicity, as observed with immune-checkpoint inhibitors or therapeutics targeting dendritic cells (DCs). Next to all these factors, there might still be unknown ones as well contributing to unwanted immunogenicity. Together, this illustrates the complexity of the question.
Why use an in vitro immunogenicity assessment?
FDA guidance suggests that in vivo models are not predictive for immunogenicity in humans; thus in vitro models using innate and adaptive immune cells are a more viable option.1
To address the different steps in the immune answer cascade, the use of in silico and in vitro models using innate and adaptive immune cells isolated from healthy donors are a more viable option, as an animal model has reduced predictive value for human immunogenicity. Although the pre-clinical risk analyses performed with these in silico and in vitro tools are not mandatory, they add a valuable quality check in the development program of the biotherapeutic.2 They can help pharmaceutical and biotech companies avoiding taking forward a candidate with a high immunogenic risk potential. Many efforts are put in harmonizing these assays amongst industry, to facilitate comparison amongst them.2
- Guidance for Industry. Immunogenicity Assessment for Therapeutic Protein Products. US Department of Health and Human Services. Food and Drug Administration. Center for Drug Evaluation and Research (CDER). Center for Biologics Evaluation and Research (CBER). August 2014. Available at: https://www.fda.gov/media/85017/download
- Assay format diversity in pre-clinical immunogenicity risk assessment: Toward a possible harmonization of antigenicity assays - PubMed (nih.gov)