Companion Diagnostics (CDx)
Clinical Trial and Clinical Diagnostic Solutions - Fulfilling the promise of personalized medicine through Companion Diagnostics
Companion Diagnostics (CDx)
Clinical Trial and Clinical Diagnostic Solutions - Fulfilling the promise of personalized medicine through Companion Diagnostics
Anatomic Pathology Laboratory Services
Bioanalytical Services
ADME / DMPK Services
Biomarker Services
Biotech Laboratory Solutions
Central Laboratory Services
Clinical Genomics Laboratory Services
Companion Diagnostics
COVID-19 Clinical Trial Solutions
Flow Cytometry Services
Translational Science and Innovations Laboratory (TSAIL)
Vaccine Laboratory Services
A laboratory partner with global experience for your CDx development
The improvements precision medicine brings to patient outcomes are both well-known and implemented in clinical practice. Companion Diagnostics (CDx) are assays that are required to be used before a specific therapy can be initiated. They are often co-developed alongside a therapeutic during its clinical development journey. Extensive testing in clinical trials is required, often in multiple sites across the globe. In fact, global solutions are needed due to the increase in clinical development in China and the challenging regulatory conditions on sample import and export. We have supported CDx assays for more than 12 years, and continually enhances its technology, bioinformatics, regulatory, clinical and commercialization aspects. With extensive experience, our organization has the appropriate structure and attributes to help deliver successful CDx programs.
Extensive CDx track record
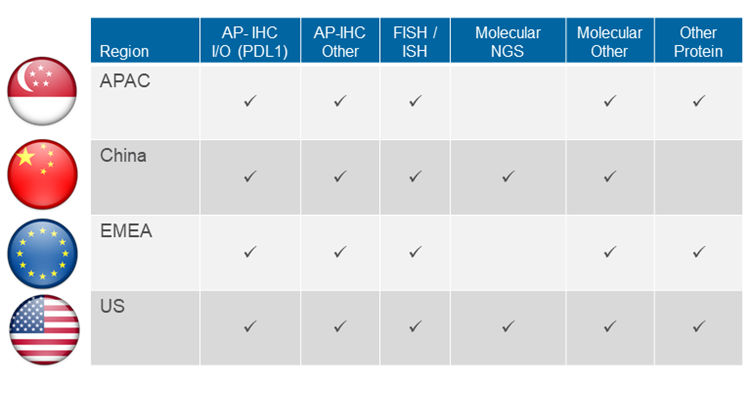
We have extensive companion diagnostics experience across the world, including APAC, China, EMEA and US, and we have generated lab data supporting multiple successful CDx submissions and more than 150 CDx clinical trials engagements.Early engagement with our experts can help circumvent issues during clinical development and provide a line of sight for commercial/post approval assay testing. Early in the co-development process, our experts lead discussions on key topics, such as assay validation versus its intended use and the regulatory paths to enable approval.
Technology breadth for companion diagnostics services
With an extensive instrument footprint in laboratories across the world, we can service CDx requests for protein expression, gene expression, mutation detection, copy number and translocations. Our current experience and focus are with but not limited to following technologies:
| Technology | Description |
| Immunohistochemistry (IHC) | IHC continues to be a dominant technology |
| Next Generation Sequencing (NGS) | Tumor Mutational Burden (TMB) used via relationship with partner Illumina using TSO 500 NGS gene panel |
| Quantitative PCR (qPCR) | This is still used; there is movement away from binary biomarkers towards panels |
A globally harmonized footprint
Most clinical development programs require testing to occur in sites across the globe, and we have harmonized CDx SOPs. Our on-site pathologists are trained across indications and IHC cut-off evaluations by the IVD company for IHC cut-off evaluations, and we dedicate CDx study coordinators and both regional and local project managers on every CDx study. Also, all pathologists are Q2 Solutions employees.
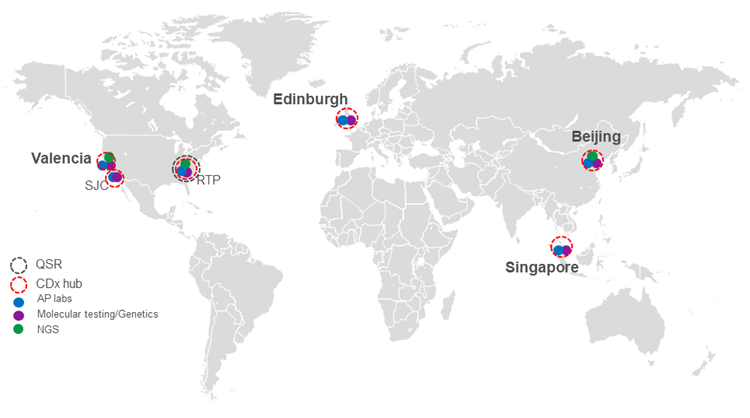
- Principal Investigators at each wholly-owned facility
- CDx Training and Harmonized SOPs across global sites
- Global logistics network to route samples into CDx labs for rapid TAT
We work with IVD companies who provide developed assays that we test at our central labs, following verification of performance, and our central labs are qualified by IVD partner companies. In addition, following Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification, utilizing a central laboratory that has been previously qualified allows the process to be streamlined through a site initiation visit (SIV) and proficiency training and study start-up.
Principal investigators and study coordinators ensure both compliance to the testing and regulatory requirements, and operational excellence through the delivery of data to clinical sites and study sponsors.
CDx solutions: alternative CDx models and paths to approval
CDx projects are often complex, typically involving an in-vitro diagnostic (IVD) company, a global central laboratory and pharmaceutical company. These projects are not without substantial cost and risk in their development. Accordingly, being able to provide different business and execution models can help circumvent some of this risk.We support the traditional path of CDx development through our network of global laboratories by testing samples using IVD products. Concurrently, as an alternative, a single-site premarket approval (ssPMA) model is increasingly considered within the industry. This model is appealing in specific circumstances, and a critique of the technical, regulatory and commercial requirements by our CDx experts can direct co-development strategies towards the most appropriate route.
As part of the CDx assay development strategy, it is good practice to review both options with a line of sight towards the ultimate positioning of the assay in the marketplace. Many considerations will determine the best path. From our experience, we are able to provide recommendations for a traditional path to CDx development or a ssPMA path that could be supported under our QSR infrastructure.
Q2 Solutions has worked on >150 CDx studies
Related Services & Solutions
Related Thought Leaders Insights
The potential of applying Flow Cytometry as an IVD technology for CDx
In this episode, Megan McCausland, Scientific Advisor for Flow Cytometry at Q2 Solutions, and Scott Bornheimer, Associate Director of Medical and Scientific Affairs at BD Biosciences, discuss the...
Companion Diagnostics Expertise
Companion diagnostics (CDx) enable personalized medicine by identifying likely patient responders based on efficacy and safety. Guidance recommends that sponsors consider CDx early in drug...
Successful Companion Diagnostics Programs: Experiences of a Global Central Laboratory
This presentation reviews the central laboratory’s role and best practices for executing companion diagnostics globally. It focuses on key attributes required for complex oncology trials, and...